GenEmbryomics
Private
Company Overview
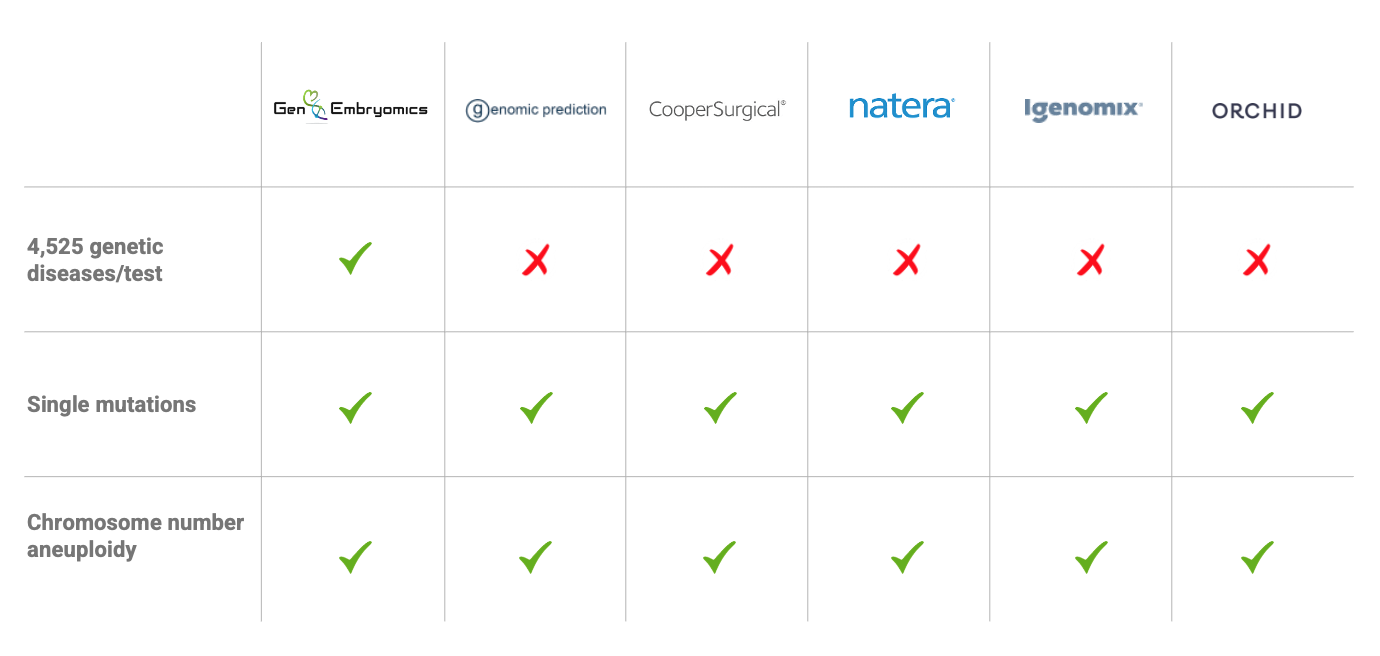
GenEmbryomics is the first company in the world to offer a comprehensive whole-genome sequencing screening test for in vitro fertilization (IVF) embryos, providing a novel way to screen for genetic diseases before pregnancy. De novo mutations spontaneously occurring at fertilization account for over 4,525 genetic diseases that are routinely missed by currently used preimplantation genetic testing.

Value Proposition
GenEmbryomics has developed the world’s first genetic screening for IVF capable of screening for more than 4,500 genetic conditions. GenEmbryomics innovative screening solution is comparatively priced to current testing solutions which have limited screening capabilities.
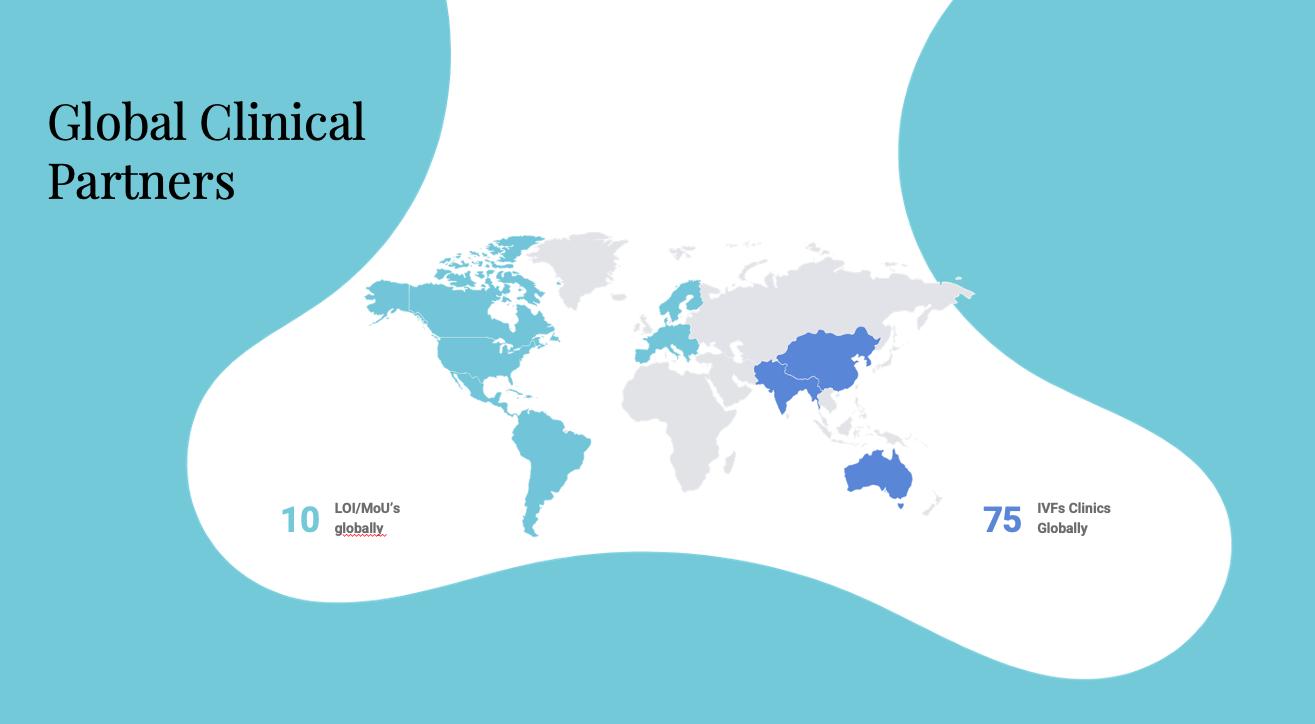
The Company is employing a B2B business model targeting IVF clinics globally. Initial commercialization plans are focused on the US, Europe, and Middle East, leveraging the key opinion leader status of the Company’s Executive Director and Senior Advisor, Dr. Santiago Munné. Dr. Munné developed the first Preimplantation Genetic Diagnosis (PGD) test to detect embryonic numerical chromosome abnormalities to avoid Down’s syndrome and other abnormalities and is extremely well-known amongst clinic owners.
Globally, the IVF market was estimated at $16.9 billion in 2018 and is expected to grow to $36.4 billion in 2026, a compound annual growth rate of 10.1%. GenEmbryomics is ideally positioned to capitalize on this demand, creating a compelling opportunity for early investors.
Genomescreen® Vs. Competitors
Investment Highlights
-
Novel solutions addressing a multibillion-dollar market opportunity
- Developed world’s first genetic screening for IVF capable of screening 4,500+ genetic conditions
- Global IVF market estimated at $16.9 billion in 2018 and expected to surpass $36 billion by 2026
- 300,000 IVF cycles performed in US annually
-
Near-term commercialization
- B2B model targeting IVF clinics
- Class II medical device with no clinical or pre-clinical studies required
- 10 LOIs/MOUs signed in US
- Excellent reimbursement potential
-
Intellectual Property
- US and Japan (70% of global market), China (fastest growing market globally), EU & Australia
-
Highly experienced executive team
- Nick Murphy, PhD – CEO/Founder: Co-inventor of the COVID-19 Risk test and Polygenic Risk Multi-Test (Genetic Technologies). First to directly sequence the human HLA system from a single chromosome.
- Santiago Munné, PhD – Executive Director & Senior Advisor: PhD in Human Genetics (University of Pittsburgh). CIO at Overture Life. Founder of: Reprogenetics, Recombine, Phosphorus, MedAnswers, Overture Life, G1 Sciences. Advisory Board (currently): Overture Life, Phosphorus, MedAnswers, Butterfly & Sama
- Monica Petica, MSC – Chief Clinical Officer: Monash IVF, Victorian Clinical Genetics Services (VCGS), Australian Forensic Police Services (Victoria, South Australia, Northern Territory)



