OxySure Systems
(OTCQB: OXYS)
RESEARCH REPORT
November 17, 2015
Stock Information
Market Data
| Fiscal Year | December |
| Industry | Medtech |
| Market Cap | $6.2M |
| Price/Earning (ttm) | N/A |
| Price/Book (mrq) | 2.6x |
| Price/Sales (ttm) | 1.8x |
| Insider Ownership | 46.1% |
| Shares Outstanding | 36.2M |
| Equity Float | 13.5M |
| Avg. Volume (3 mo.) | 146,786 |
Income Statement Snapshot
| Revenue (TTM) | $3.4M |
| Gross Profit (TTM) | $1.4M |
| Gross Margin (TTM) | 42.5% |
| Net Income (TTM) | ($5.7M) |
Balance Sheet Snapshot
| Cash (MRQ) | $2.0M |
| Debt (MRQ) | $1.2M |
Research Update
3Q15 Revenue Up 39% YoY and 9% QoQ; 14™ Straight Qtr of Revenue Growth
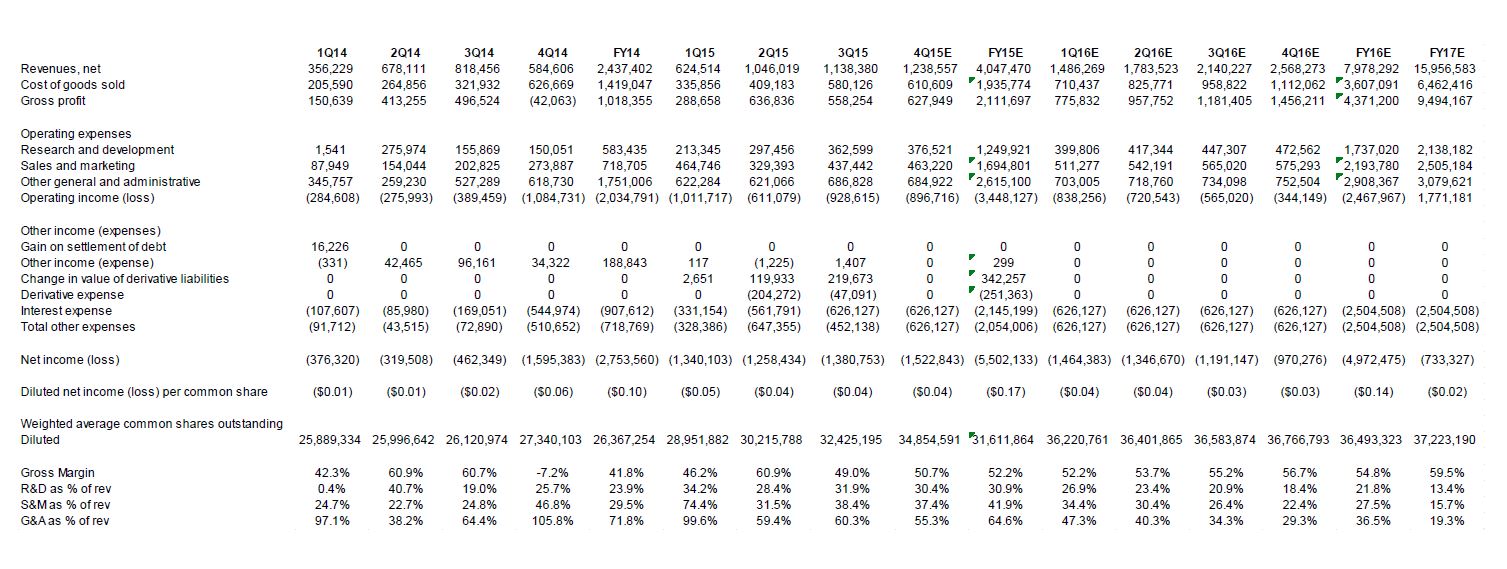
3Q15 revenue up 39% YoY and 9% QoQ; 14™ straight quarter of growth. OXYS reported revenue of $1.1 million in 3Q15, a 39% YoY increase and 9% QoQ increase as compared to $818,456 in 3Q14 and $1.0 million in 2Q15. Gross profit was $558,254 (gross margin of 49.0%), a 12% YoY increase from $496,524 in 3Q14 (gross margin of 60.7%). Selling, general and administrative expenses for 3Q15 were $1.5 million as compared to $885,983 in 3Q14. The primary reasons for the expense increases were investments in sales and marketing (a 116% YoY increase to $437,442), and an increase in research and development (a 133% YoY increase to $362,599). We expect the Company to continue to invest heavily in sales and marketing and research and development as the Company expands its direct sales force and invests in new product development. Net loss for 3Q15 was $(1.4 million), or $(0.04) per share, as compared to $(462,349) or $(0.02) per share in 3Q14.
The Company expects to reach a $10 million revenue run rate in 2016, along with reaching cash flow breakeven and GAAP breakeven (with cash flow breakeven occurring first). Also, the Company has a growing sales pipeline of $3.5 million. OXYS anticipates closing on approximately 70%-80% of this pipeline, and the time to close is expected to show additional improvement as the Company improves its sales technique with experience. Additonally, OXYS has stated that it has seen a significant uptick in sales to law enforcement agencies.
New product additions expected to increase sales; already seeing strong demand from Quickclot and AED sales.OXYS has seen significant sales and demand from its additions of the Quickclot Hemostatic Solution and the new Cardiac Science Powerheart® G5 AED. We expect these products to increase OXYS’s sales further in the coming months.
Journal of Emergency Medicine (JEMS) opinion paper leads to increased awareness and support for “public/first aid oxygen” usage. JEMS, a widely respected medical journal, recently published an opinion piece on the usability of “public/first aid oxygen” usage, prior to the arrival of professional first responders on the scene. The article also had commentary from Pete Goldman MD, an emergency department physician who has been an innovator in public involvement in emergency care delivery for decades. The opinion piece concluded that FDA-approved stock first aid oxygen units, with flow rates of 6-7 LPM, are probably safe to use without pulse oximetry. The article provides additional evidence that there are likely not any concerns about “over-oxygenation,” and that FDA-approved oxygen units are safe to be administered by individuals before the arrival of trained first responders.
Having an opinion piece published by a respected journal such as JEMS will likely convince many more organizations to purchase the OxySure Model 615, due to the addition of a very respected, third-party medical opinion. This could lead to a strong increase in sales as this paper becomes known by end user groups.
Price target of $1.35, which represents EV/S multiples of 11.9x FY15E revenue, 6.0x FY16E revenue, and 3.0x FY17E revenue.
Additional Information
Auditor: Sadler, Gibb & Associates, LLC
Legal Counsel: K&L Gates, The Law Firm of Gregory G. Jones, P.C., The Johnson Firm, Horzepa Spiegel & Associates, PC
Transfer Agent: Action Stock Transfer
OxySure Systems, Inc.
10880 John W. Elliott Drive, Suite 600,
Frisco, TX 75033
+1 (972) 294 6461
Investor Contact Info:
RedChip Companies, Inc.
1017 Maitland Center Commons Blvd.
Maitland, FL 32751
(407) 644-4256
www.redchip.com




